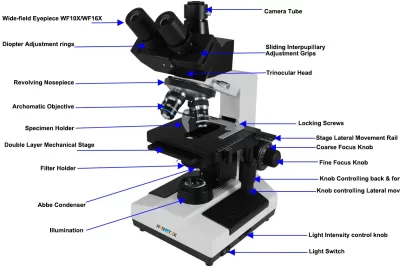
What chemical properties influence poisoning and pollution

Poisoning and environmental pollution remain critical challenges in today's society, largely driven by the chemical properties of various substances. Understanding how these chemical properties influence both toxicity and pollutant behavior is essential for mitigating risks and protecting public health. With the rise of industrialization and urbanization, the release of harmful chemicals like carbon monoxide (CO) into the environment has become a focal point of research and legislation.
One of the most concerning aspects of chemical pollution is its widespread impact on ecosystems and human health. Toxic substances can have devastating effects, leading to serious health conditions or even death. This article aims to explore the chemical properties that contribute to poisoning and environmental damage, focusing on CO formation, the mechanisms of exposure, and strategies for treatment and remediation. By dissecting these elements, we hope to shed light on the intricate relationship between chemical properties and their implications for safety and environmental integrity.
- Overview of Chemical Properties
- The Role of Toxicity in Poisoning
- Mechanisms of Exposure and Absorption
- Environmental Impact of Chemical Pollutants
- Chemical Reactions Contributing to Pollution
- Carbon Monoxide as a Case Study
- Identifying and Mitigating Risks
- Treatment and Remediation Strategies
- Conclusion
Overview of Chemical Properties
The behavior of chemicals is largely determined by their chemical properties, which include characteristics like solubility, reactivity, and molecular structure. These properties dictate how substances interact with each other and their environment, ultimately influencing their potential for toxicity and pollution.
Physical and Chemical Characteristics
Every chemical has unique physical properties such as boiling point, melting point, and state at room temperature, which can determine how they exist in the environment. For instance, carbon monoxide (CO) is a gas at room temperature, with a melting point of -205°C and a boiling point of -191.5°C. In terms of its chemical characteristics, CO is relatively stable under standard conditions, but it readily reacts with other substances at elevated temperatures.
Acidity and Basicity
The pH level of a substance can also play a crucial role in its chemical behavior. Acidic or basic substances can lead to various environmental issues, from soil degradation to water contamination. Understanding how these properties influence the interaction of chemicals can help in predicting their potential adverse effects.
The Role of Toxicity in Poisoning
Toxicity is a measure of the adverse effects of a chemical on living organisms, and it is influenced by factors such as dose, duration of exposure, and individual susceptibility. The chemical structure of a toxin determines its mode of action, including how it interacts with biological systems to induce harm.
Types of Toxicity
Toxicity can be categorized into acute and chronic forms. Acute toxicity results from short-term exposure to high levels of a substance, while chronic toxicity arises from ongoing exposure to lower levels over long periods. In both cases, understanding the chemical properties of a toxin is crucial for determining its potential health impacts.
Variables Influencing Toxicity
Numerous chemical factors influence toxicity, including molecular weight, solubility, and the presence of functional groups. Substances with low molecular weights and high solubility often have increased bioavailability, leading to higher toxicity levels. Additionally, some chemicals can form metabolites, which may be even more toxic than their parent compounds.
Mechanisms of Exposure and Absorption
Exposure to toxic chemicals like CO can occur through various routes—ingestion, inhalation, and dermal contact. Understanding how these mechanisms work is vital for developing effective prevention and treatment strategies.
Routes of Exposure
Inhalation is the primary route for gases such as CO, which can enter the bloodstream quickly through the lungs. Once in the body, CO binds to hemoglobin with a much greater affinity than oxygen, forming carboxyhemoglobin and impeding normal oxygen transport. This binding effect is crucial in understanding the toxicological mechanisms of CO poisoning.
Factors Affecting Absorption
Several factors influence the absorption of toxic substances, including physical state (solid, liquid, gas), particle size, and formulation. For instance, particulate matter can have a higher impact due to its ability to penetrate deep into the respiratory tract. In the case of CO, even small concentrations can lead to significant binding to hemoglobin, highlighting the importance of its chemical properties in toxic outcomes.
Environmental Impact of Chemical Pollutants
Chemical pollutants can have a lasting impact on the environment, altering ecosystems and harming wildlife. The persistence and mobility of these chemicals are largely governed by their chemical properties.
Persistence in the Environment
Some chemicals can persist in the environment for long periods, contributing to ongoing pollution problems. For example, heavy metals and certain organic pollutants can remain in soil and water, leading to bioaccumulation in living organisms. Understanding the persistence of different chemicals helps in assessing their long-term environmental impact.
Bioaccumulation and Biomagnification
Bioaccumulation occurs when organisms absorb toxic substances faster than they can eliminate them, while biomagnification refers to the increasing concentration of toxins in organisms at higher levels of the food chain. The chemical properties of a pollutant, such as solubility and resistance to degradation, determine its potential for bioaccumulation.
Chemical Reactions Contributing to Pollution
Chemical reactions can lead to the formation of pollutants in several ways. Understanding these reactions provides insight into how to mitigate their effects.
Combustion and Incomplete Combustion
Incomplete combustion of carbon-based fuels results in the production of harmful byproducts like carbon monoxide (CO) and particulate matter. Effective combustion control measures are essential for reducing the environmental impact of these emissions, highlighting the importance of understanding the chemical reactions involved.
Secondary Reactions in the Atmosphere
Certain pollutants can undergo secondary reactions in the atmosphere, resulting in the formation of secondary pollutants like ozone. These reactions depend on the presence of various catalysts and environmental conditions. Understanding these interactions is critical for forming effective environmental policies.
Carbon Monoxide as a Case Study
Carbon monoxide (CO), a colorless and odorless gas, serves as an important case study in understanding chemical properties related to poisoning and pollution.
Production and Sources
CO is primarily produced by the incomplete combustion of organic materials, such as fossil fuels. Major sources include motor vehicle exhaust, residential heating, and industrial processes. Its notable chemical properties make it a common environmental pollutant that poses significant health risks.
Toxicity and Health Effects
The toxicity of CO arises from its ability to bind to hemoglobin in red blood cells, creating carboxyhemoglobin and reducing oxygen delivery to tissues. Symptoms of CO poisoning can range from headaches and dizziness to loss of consciousness and even death, depending on exposure levels. Immediate intervention is crucial in cases of CO poisoning, typically involving oxygen therapy to displace CO from hemoglobin.
Identifying and Mitigating Risks
Recognizing the potential risks associated with chemical exposure is critical for public safety. Awareness, proper monitoring, and preventive strategies can significantly reduce the likelihood of poisoning incidents.
Monitoring and Detection
Utilizing advancements in technology for the detection of harmful pollutants can aid in effectively managing chemical exposure. Continuous monitoring of air quality, particularly in industrial areas, is essential for identifying sources of CO and other pollutants.
Preventive Measures
Establishing strict regulations and implementing safety protocols can greatly reduce exposure risks. For example, improving combustion efficiency and upgrading ventilation systems can substantially lower CO emissions in residential and industrial settings.
Treatment and Remediation Strategies
Effective treatment and remediation strategies are essential to respond to poisoning incidents and environmental contamination.
Medical Treatment of CO Poisoning
The primary treatment for CO poisoning involves oxygen therapy, which helps to displace CO from hemoglobin. In more severe cases, hyperbaric oxygen therapy may be employed. Understanding the chemical properties of CO is vital in guiding appropriate medical responses and improving outcomes for affected individuals.
Remediation of Contaminated Environments
Methods for remediating contaminated sites may include chemical, biological, or physical treatments designed to neutralize or remove pollutants. An understanding of the specific chemical properties of contaminants informs the selection of the most effective remediation strategy.
Conclusion
Understanding the chemical properties that influence poisoning and pollution is imperative for addressing current environmental and health challenges. As illustrated through the case study of carbon monoxide, the interplay between chemical characteristics and their environmental impact is complex but critical. By promoting awareness, improving detection methods, and implementing effective treatment and remediation strategies, we can better navigate the risks associated with toxic substances. Ultimately, advancing our understanding of these chemical properties will pave the way for enhanced safety and environmental stewardship.
Did you find this article helpful? What chemical properties influence poisoning and pollution See more here Education.
Leave a Reply


Related posts